
Our research
For several years, our laboratory has been interested in the mechanisms that control mitotic entry, progression and exit. The main aim of mitosis, a crucial phase of the cell cycle, is to assure equal chromosome segregation in divided cells. For that, an extensive number of mitotic regulators are activated, re-localized, modified, degraded… These modifications are under the control of several signalling pathways that modulate the different phases of mitosis. We focus our interest on the molecular mechanisms that control cell division. Notably, we are interested in the coordinated activation/inactivation of multiple kinases and phosphatases and their substrates that, together, ensure the correct implementation of mitosis and meiosis. Our recent results show that these mitotic regulators could also play a role in the control of other cell cycle phases or of other cell features, such as DNA replication or cell transformation.
Read more
The Greatwall/ARPP19-ENSA/PP2A-B55 pathway and the cell cycle.
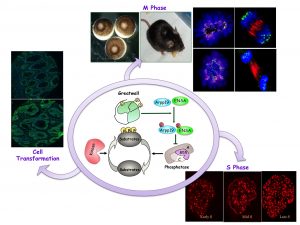
Our laboratory studies the role of kinases and phosphatases in the regulation of mitotic entry and progression. Mitosis requires massive protein phosphorylation that is mostly promoted by the master kinase cyclin B-CDK1. Historically, it was assumed that protein phosphorylation in mitosis was under the control of cyclin B-CDK1 and that the phosphatase activity responsible of protein dephosphorylation was constant. For the first time, we demonstrated that like for the kinase activity, phosphatase activity is also modulated for correct mitosis. Our studies in Xenopus laevis egg extracts identified a new pathway that controls mitotic phosphatase activity and that includes the kinase Greatwall (GWL) and its substrates ARPP19 and ENSA. We showed that at mitotic entry, GWL is activated and phosphorylates ARPP19 and ENSA, thus promoting their binding to PP2A-B55 and the inhibition of this phosphatase. This leads to stable protein phosphorylation and mitotic entry. We then demonstrated that this pathway is conserved in human cells in culture. We are currently investigating the role of GWL/ARPP19-ENSA/PP2A-B55 in tissue homeostasis by using inducible knock out mouse models. Interestingly, besides the role in mitotic division, we demonstrated that this signalling pathway is also involved in other cell cycle phases. Specifically, our recent data show that this cascade contributes to the control of S phase, by regulating DNA replication dynamics. Finally, we identified GWL as a potent oncogene that promotes cell transformation by controlling AKT activity. Next, we will try to identify the mechanisms beneath the phenotypes observed in adult knock out mice and the pathway(s) responsible of GWL-dependent AKT regulation. Moreover, we will investigate the pathway by which GWL/ENSA/PP2A-B55 controls S phase and try to identify new GWL substrates.
Funding
ANR JCJC MTDISco (01/10/2020-31/12/2025)
ANR GREATOBTREAT (01/02/2024-31/01/2029)
ANR MITODISSECT (01/11/2022-31/10/2025)
ARC (4ème année de thèse)
ARC ( 01/01/2024-31/12/2025)
Publications
2026
- Greatwall depletion from Xenopus oocytes reveals a key role of the cyclin B/CDK1-PP2A-B55 balance in the coordination of meiotic events. Sylvain Roque , Célia Ben Choug, Cedric Hassen Khodja , Suzanne Vigneron, Véronique Legros, Guillaume Chevreux, Benjamin Lacroix, Anna Castro and Thierry Lorca. Nature Communication (2026) PMID: 42129183 DOI: 10.1038/s41467-026-73011-5
- Molecular basis for the activation of Aurora A and Plk1 kinases during mitotic entry. Anaïs Pillan, Philippine Ormancey, Celia Ben Choug, Stephen Orlicky, Nicolas Tavernier , Lucie Van Hove, Batool Ossareh-Nazari, Nicolas Joly, Frank Sicheri , Thierry Lorca and Lionel Pintard. EMBO Journal (2026) PMID: 41606196 PMCID: PMC12953601 DOI: 10.1038/s44318-025-00679-8
- Inactivation of Focal Adhesion Kinase FAK Rapidly Abrogates Keratinocyte Entry in Mitosis via Rho-Associated Kinase, Resulting in Squamous Differentiation. Lizbeth Contreras, Lorena García-Gaipo, Fe García-Reija, Anna Castro, Juana M García-Pedrero, Thierry Lorca and Alberto Gandarillas. FASEB Journal (2026) PMID: 41521614 DOI: 10.1096/fj.202500347R
- FAM122A inhibition of PP2A-B55 through a bipartite binding mechanism. Iker Benavides-Puy, Suzanne Vigneron, Arminja N Kettenbach, Thierry Lorca and Jakob Nilsson. Journal of Cell Science In press (2026)
2025
- The MAST kinase KIN-4 carries out mitotic entry functions of Greatwall in C. elegans. Roumbo L, Ossareh-Nazari B, Vigneron S, Stefani I, Van Hove L, Legros V, Chevreux G, Lacroix B, Castro A, Joly N, Lorca T, Pintard L. EMBO J. 2025 Feb 17. Pubmed
2024
- Measuring Mitotic Spindle and Microtubule Dynamics in Marine Embryos and Non-model Organisms. Chenevert J, Robert MLV, Sallé J, Cacchia S, Lorca T, Castro A, McDougall A, Minc N, Castagnetti S, Dumont J, Lacroix B. Methods Mol Biol. 2024;2740:187-210. Pubmed
- Cell Cycle Control : Methods and Protocols. Castro A & Lacroix B. Editors Part of the book series: Methods in Molecular Biology (Springer Protocols, Humana Press. 2024. Link
- Increases in cyclin A/Cdk activity and in PP2A-B55 inhibition by FAM122A are key mitosis-inducing events. Lacroix B, Vigneron S, Labbé JC, Pintard L, Lionne C, Labesse G, Castro A, Lorca T. EMBO J. 2024 Feb 20. Pubmed
2023
- Synergistic stabilization of microtubules by BUB-1, HCP-1 and CLS-2 controls microtubule pausing and meiotic spindle assembly. Macaisne N, Bellutti L, Laband K, Edwards F, Pitayu-Nugroho L, Gervais A, Ganeswaran T, Geoffroy H, Maton G, Canman JC, Lacroix B, Dumont J. Elife. 2023 Feb 17;12:e82579. Pubmed
2022
- Structural organization and dynamics of FCHo2 docking on membranes. El Alaoui F, Casuso I, Sanchez-Fuentes D, Arpin-Andre C, Rathar R, Baecker V, Castro A, Lorca T, Viaud J, Vassilopoulos S, Carretero-Genevrier A, Picas L. Elife. 2022 Jan 19;11:e73156. Pubmed
- PP2A-B55: substrates and regulators in the control of cellular functions. Amin P, Awal S, Vigneron S, Roque S, Mechali F, Labbé JC, Lorca T, Castro A. Oncogene. 2022 Jan;41(1):1-14. Pubmed
2021
- The study of the determinants controlling Arpp19 phosphatase-inhibitory activity reveals an Arpp19/PP2A-B55 feedback loop. Labbé JC, Vigneron S, Méchali F, Robert P, Roque S, Genoud C, Goguet-Rubio P, Barthe P, Labesse G, Cohen-Gonsaud M, Castro A, Lorca T. Nat Commun. 12(1):3565. Pubmed
- Bora phosphorylation substitutes in trans for T-loop phosphorylation in Aurora A to promote mitotic entry. Tavernier N, Thomas Y, Vigneron S, Maisonneuve P, Orlicky S, Mader P, Regmi SG, Van Hove L, Levinson NM, Gasmi-Seabrook G, Joly N, Poteau M, Velez-Aguilera G, Gavet O, Castro A, Dasso M, Lorca T, Sicheri F, Pintard L. Nat Commun. 12(1):1899 Pubmed
2020
- PP2A-B55 Holoenzyme Regulation and Cancer. Perrine Goguet-Rubio, Priya Amin, Sushil Awal, Suzanne Vigneron, Sophie Charrasse, Francisca Mechali, Jean Claude Labbé, Thierry Lorca, Anna Castro. Biomolecules . 10(11):1586. Pubmed
2019
- Arpp19 and ENSA, two PP2A-B55 phosphatase inhibitors that differentially control the cell cycle. Hached K, Goguet-Rubio P, Charrasse S, Lorca T, Castro A. Med Sci (Paris). 35(6-7):504-506 Pubmed
- Nuclear import of Xenopus egg extract components into cultured cells for reprogramming purposes: a case study on goldfish fin cells. Chênais N, Lorca T, Morin N, Guillet B, Rime H, Le Bail PY, Labbé C. Sci Rep. 9(1):2861. Pubmed
- ENSA and ARPP19 differentially control cell cycle progression and development. Hached K, Goguet P, Charrasse S, Vigneron S, Sacristan MP, Lorca T, Castro A. J Cell Biol. pii: jcb.201708105. Pubmed
2018
- Greatwall kinase at a glance. Castro A, Lorca T. J Cell Sci. 131(20). Pubmed
- Cyclin A-cdk1-Dependent Phosphorylation of Bora Is the Triggering Factor Promoting Mitotic Entry. Vigneron S, Sundermann L, Labbé JC, Pintard L, Radulescu O, Castro A, Lorca T. Dev Cell. 45(5):637-650. Pubmed
2017
- Ensa controls S-phase length by modulating Treslin levels. Charrasse S, Gharbi-Ayachi A, Burgess A, Vera J, Hached K, Raynaud P, Schwob E, Lorca T, Castro A. Nat Commun. 8(1):206 Pubmed
- Loss of Centromere Cohesion in Aneuploid Human Oocytes Correlates with Decreased Kinetochore Localization of the Sac Proteins Bub1 and Bubr1. Lagirand-Cantaloube J, Ciabrini C, Charrasse S, Ferrieres A, Castro A, Anahory T, Lorca T. Sci Rep. 7:44001. Pubmed
2016
- CDK1 Prevents Unscheduled PLK4-STIL Complex Assembly in Centriole Biogenesis. Zitouni S, Francia ME, Leal F, Montenegro Gouveia S, Nabais C, Duarte P, Gilberto S, Brito D, Moyer T, Kandels-Lewis S, Ohta M, Kitagawa D, Holland AJ, Karsenti E, Lorca T, Lince-Faria M, Bettencourt-Dias M. Curr Biol. 26:1127-37. Pubmed
- The master Greatwall kinase, a critical regulator of mitosis and meiosis. Vigneron S, Robert P, Hached K, Sundermann L, Charrasse S, Labbé JC, Castro A, Lorca T. Int J Dev Biol. 60:245-254. Pubmed
- Greatwall dephosphorylation and inactivation upon mitotic exit is triggered by PP1. Ma S, Vigneron S, Robert P, Strub JM, Cianferani S, Castro A, Lorca T. J Cell Sci. 129:1329-39. Pubmed
2015
- Greatwall promotes cell transformation by hyperactivating AKT in human malignancies. Vera J, Lartigue L, Vigneron S, Gadea G, Gire V, Del Rio M, Soubeyran I, Chibon F, Lorca T, Castro A. 4:e10115. Pubmed
- Global Phosphoproteomic Mapping of Early Mitotic Exit in Human Cells Identifies Novel Substrate Dephosphorylation Motifs. McCloy RA, Parker BL, Rogers S, Chaudhuri R, Gayevskiy V, Hoffman NJ, Ali N, Watkins DN, Daly RJ, James DE, Lorca T, Castro A, Burgess A. Mol Cell Proteomics. 14:2194-212. Pubmed
2014
- Partial inhibition of Cdk1 in G 2 phase overrides the SAC and decouples mitotic events. McCloy RA, Rogers S, Caldon CE, Lorca T, Castro A, Burgess A. Cell Cycle. 13:1400-12. Pubmed
2013
- Greatwall is essential to prevent mitotic collapse after nuclear envelope breakdown in mammals. Álvarez-Fernández M, Sánchez-Martínez R, Sanz-Castillo B, Gan PP, Sanz-Flores M, Trakala M, Ruiz-Torres M, Lorca T, Castro A, Malumbres M. Proc Natl Acad Sci U S A. 110(43):17374-9. Pubmed
- Budding yeast greatwall and endosulfines control activity and spatial regulation of PP2A(Cdc55) for timely mitotic progression. Juanes MA, Khoueiry R, Kupka T, Castro A, Mudrak I, Ogris E, Lorca T, Piatti S. PLoS Genet. 9(7):e1003575. Pubmed
2012
- Quantitative live imaging of endogenous DNA replication in mammalian cells. Burgess A, Lorca T, Castro A. PLoS One. 7(9):e45726. Pubmed
- Deciphering the New Role of the Greatwall/PP2A Pathway in Cell Cycle Control. Lorca T, Castro A. Genes Cancer. 3(11-12):712-20. Pubmed
- CDK-dependent potentiation of MPS1 kinase activity is essential to the mitotic checkpoint. Morin V, Prieto S, Melines S, Hem S, Rossignol M, Lorca T, Espeut J, Morin N, Abrieu A. Curr Biol. 22(4):289-95. Pubmed
2011
- Characterization of the mechanisms controlling Greatwall activity. Vigneron S, Gharbi-Ayachi A, Raymond AA, Burgess A, Labbé JC, Labesse G, Monsarrat B, Lorca T, Castro A. Mol Cell Biol. 31(11):2262-75. Pubmed
- [Greatwall, a new guardian of mitosis].[Article in French] Gharbi-Ayachi A, Burgess A, Vigneron S, Labbé JC, Castro A, Lorca T. Med Sci (Paris). 27(4):352-4. Pubmed
2010
- Loss of human Greatwall results in G2 arrest and multiple mitotic defects due to deregulation of the cyclin B-Cdc2/PP2A balance. Burgess A, Vigneron S, Brioudes E, Labbé JC, Lorca T, Castro A. Proc Natl Acad Sci U S A. 107(28):12564-9. Pubmed
- Constant regulation of both the MPF amplification loop and the Greatwall-PP2A pathway is required for metaphase II arrest and correct entry into the first embryonic cell cycle. Lorca T, Bernis C, Vigneron S, Burgess A, Brioudes E, Labbé JC, Castro A. J Cell Sci. 123(Pt 13):2281-91. Pubmed
- The substrate of Greatwall kinase, Arpp19, controls mitosis by inhibiting protein phosphatase 2A. Gharbi-Ayachi A, Labbé JC, Burgess A, Vigneron S, Strub JM, Brioudes E, Van-Dorsselaer A, Castro A, Lorca T. Science. 330(6011):1673-7. Pubmed
2009
- Greatwall maintains mitosis through regulation of PP2A. Vigneron S, Brioudes E, Burgess A, Labbé JC, Lorca T, Castro A. EMBO J. 28(18):2786-93. Pubmed
Phosphatases in cell cycle and signaling

 Anna CASTRO & Thierry LORCA
Anna CASTRO & Thierry LORCA
Group leaders (Research Directors CNRS)
Team Members
(Chercheur DR1) +33 (0)4 34 35 95 56 |
|
(Chercheur DR1) +33 (0)4 34 35 95 56 |
|
(IE-Recherche) +33 (0)4 34 35 95 56 |
|
(Chercheur) +33 (0)4 34 35 95 56 |
|
(Doctorant) +33 (0)4 34 35 95 57 |
|
(Doctorant) +33 (0)4 34 35 95 57 |
|
(Doctorant) +33 (0)4 34 35 95 56 |
|
(Stagiaire) +33 (0)4 34 35 9 |
|
(Stagiaire) +33 (0)4 34 35 9 |
|
(Stagiaire) +33 (0)4 34 35 9 |
Contact us
Replace the name and address above with that of the member to contact
firstname.name@crbm.cnrs.fr
GRANTS











